Brew log · single filing
Hold the butter: diacetyl, yeast, and the popcorn plot twist
What is diacetyl?
Diacetyl (C4H6O2) is a compound that smells and tastes strongly of butter or butterscotch candy. It belongs to a family of compounds called Vicinal Diketones (VDKs).
That acronym will come back later.
But why is it in my beer?
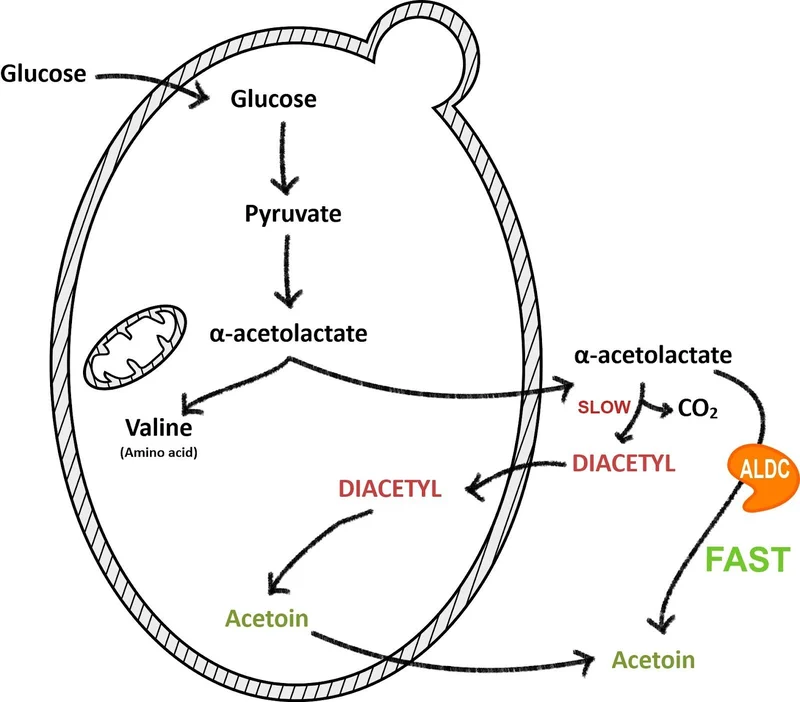
Diacetyl is a natural byproduct of fermentation.
During the early to mid stages of fermentation (the exponential growth phase), yeast synthesize the amino acid valine. One of the intermediate compounds in that process is α-acetolactate.
Some of this α-acetolactate leaks out of the yeast cell and into the beer. Once outside the cell it spontaneously converts into diacetyl.
This is why diacetyl often appears after fermentation has slowed down—because the precursor is still converting in the beer.
How do we control this?
Several factors can increase diacetyl levels in beer.
Yeast health
Unhealthy yeast require increased valine synthesis, which produces more α-acetolactate.
Low FAN in the wort
FAN (Free Amino Nitrogen) provides yeast with amino acids they need to grow. If FAN is low, yeast must synthesize more valine themselves, which increases diacetyl precursors.
Infection
Some bacteria, especially Pediococcus, can produce large amounts of diacetyl.
Also infection does lots of weird things. Come to the next club meeting and we will talk about cleaning all the things.
Ok—but if it’s natural, how do we get clean beer?
A diacetyl rest.
Near the end of fermentation, raise the temperature of the beer to 65–72°F, or at least warmer than your fermentation temperature, and hold it there for a few days.
During this stage the yeast will:
- Reabsorb diacetyl
- Convert it into acetoin
Acetoin is essentially flavor-neutral in beer.
This is exactly where we want to end up.
To recap
- Yeast produce α-acetolactate while making valine
- α-acetolactate escapes the cell
- It converts into diacetyl in the beer
- Yeast reabsorb diacetyl
- It gets converted into acetoin
Then you are done.
Are we there yet?
There is a simple test you can run called forced VDK testing (remember that acronym?).
The goal is to check whether α-acetolactate precursors are still present in the beer.
Heat accelerates the conversion of α-acetolactate into diacetyl, which makes it detectable.
The process
- Take two sealed samples of your beer. One will be the control and one will be the test.
- Mark your control sample and leave it at room temperature.
- Heat the test sample to 140–160°F for 15–30 minutes.
- Allow the heated sample to cool back to the same temperature as the control.
- Open both samples and smell them.
Interpreting the results
If both samples smell like diacetyl
You need a longer rest. Rouse the yeast if possible.
If the control is clean but the heated sample shows diacetyl
α-acetolactate is still present. Leave the beer on the yeast longer; rouse if possible.
If both samples are clean
Congratulations—you are done. No diacetyl and no remaining precursors. Package away.
Final thoughts
Diacetyl is not a brewing mistake by itself. It is a normal step in fermentation.
The trick is simply giving yeast enough time and the right conditions to clean up after themselves.
Image credit: White Labs — Compound spotlight: diacetyl